1.DCGI Enforces Revised GMP Standards (Drug Regulation)
What & Where
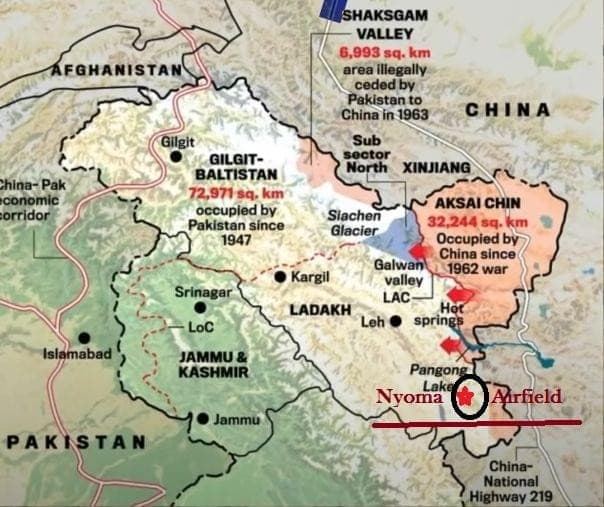
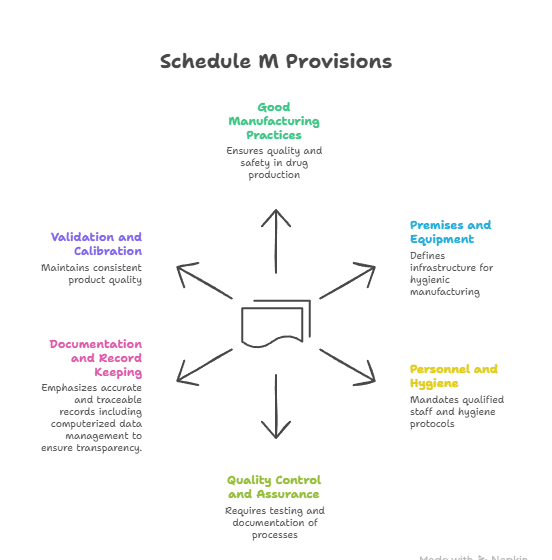
Schedule M, annex to Drugs & Cosmetics Act 1940, codifies GMP for Indian pharma manufacturing.
2023 revision renamed clause to “good manufacturing practices and requirements of plant & equipment”, mirroring WHO-GMP.
DCGI directs state regulators to enforce upgraded Schedule M nationwide by Jan 2026 after fatal cough-syrup contaminations.
Quick Facts for MCQs
Legal & Policy
- Directive; DCGI orders uniform Schedule M adoption, reducing CDSCO–state overlap.
- Provision; stresses validated processes, cleanrooms, documentation, product-recall systems.
- Aim; align with WHO-GMP, USFDA norms, restore credibility.
Quality & Safety
- Incident; contaminated paediatric syrups killed children in Gambia, Uzbekistan, India.
- Concern; repeated WHO alerts tarnish “Pharmacy of the World” image.
- Barrier; MSME units face high costs for labs, data-integrity upgrades.
Economic Angle
- Growth; pharma market projected USD 130 bn by 2030 at >10 % CAGR.
- Export; US, Belgium, S. Africa, UK, Brazil dominate destinations.
- Risk; heavy API import reliance despite PLI-backed bulk-drug parks.
Key Data Points
| Feature | Data-Point |
|---|---|
| Enforcement deadline | January 2026 |
| Supervising authority | Drugs Controller General of India |
| India’s pharma rank (volume) | 3rd globally |
| Share in global generic exports | ≈20 % |
| FY 24 drug & pharma exports | USD 27.85 bn |
| API import dependence | ~70 % (largely China) |
| UNICEF vaccine supply | 55-60 % from India |
| WHO DPT demand met | 99 % by India |
| 2024 biotech sector size | >USD 130 bn |
| Top export markets | USA, Belgium, S. Africa, UK, Brazil |
Related UPSC Prelims PYQs
Which organization developed the Online National Drugs Licensing System (ONDLS) portal?